
Rapid Pest Risk Analysis (PRA) for:
Phytophthora pluvialis
October 2022
Summary and conclusions of the rapid PRA
This rapid PRA has been undertaken following a finding of the Oomycete pathogen Phytophthora pluvialis in Cornwall in September 2021. Subsequently, additional findings have been made elsewhere in England, also in Scotland and Wales on western hemlock and Douglas fir but not in Northern Ireland. Prior to the UK detections P. pluvialis was only known to be present in New Zealand and the Pacific Northwest (PNW) of the USA. In New Zealand it causes a disease known as red needle cast, primarily on radiata pine and Douglas fir, causing premature needle loss and loss of increment growth but trees recover. In the PNW it causes very minor damage on Douglas fir or tanoak and is considered part of the native forest mycota. This PRA shows:
Risk of entry
P. pluvialis is already present in the United Kingdom, the PRA area, but with a limited distribution across the UK and is the subject of official control. With the current plant health measures that are in place the likelihood of further entry via various pathways is assessed. Entry via pathway (1) traded plants for planting is considered as very unlikely (low confidence); pathway (2) non-traded plants as unlikely (low confidence); pathway (3) timber/WPM as very unlikely (high confidence); pathway (4) cut conifer foliage as very unlikely (medium confidence); pathway (5) seeds as unlikely (low confidence); or pathway (6) soil and contaminated forestry machinery as very unlikely (low confidence).
Risk of establishment
Establishment under protection is considered very unlikely (high confidence). Phytophthora pluvialis has already established outdoors in some areas of England, Wales, and Scotland (high confidence), with natural spread via aerial spore dispersal and in water courses likely to occur at moderate pace (medium confidence). In contrast, spread in
1
trade via infected plants is considered likely to occur quickly (medium confidence), but slowly (medium confidence) through the movement of wood/logs, and only very slowly (high confidence) via cut foliage. Lack of data on the potential host range of P. pluvialis and likelihood of sporulation on different plant parts affects confidence in these ratings.
Economic, environmental, and social impact
Based on uncertainty about host range, and the contrast in symptoms on affected trees in the UK compared with those in the PNW or New Zealand, the potential economic impact of P. pluvialis is rated large (low confidence), whilst both environmental and social impacts are assessed as medium (low confidence), with low confidence rating due to lack of data.
Endangered area
Susceptible hosts and a suitable climate for disease are present throughout much of the PRA area. The most favourable climatic conditions are likely to be throughout western UK based on comparisons with New Zealand and PNW where P. pluvialis is most active.
Risk management options
Eradication efforts are judged unlikely to succeed based on the number of findings already made. Short-term options for consideration include: (1) Containment of outbreak areas and host removal on only the most severely affected sites. (2) Controlled timber movement from affected sites. (3) Monitoring tree recovery on more lightly affected sites and what triggers serious disease episodes to understand the impacts of P. pluvialis over time.
Medium to longer-term management options include: (1) Use of silvicultural treatments to reduce the likelihood of severe disease expression. (2) Deployment of disease suppression compounds which reduce disease prevalence through targeted aerial application. (3) Breeding for resistance as a long-term option for high value hosts.
Key uncertainties/topics that would benefit from further investigation
| Section of PRA | Further work needed to improve the PRA |
| Hosts (host range) | • Host range testing for susceptibility to P. pluvialis including UK grown Pinus species and other conifer species. • Testing non-conifer species including species within the Fagaceae and some ornamental species. • Assessing recovery potential and/or mortality rates of infected trees. |
| Pathogen behaviour | • Determining the rate of spread of the pathogen through epidemiological modelling, and climate matching to identify parts of the UK most at risk and inform future planting plans. • Potential for persistence in soil and plant parts. • Sporulation potential on UK grown hosts and optimum/minimum conditions for sporulation. |
| Pathogen genetics | • Genotype comparisons of UK, NZ and USA P. pluvialis populations. • Potential for hybridisation with other Phytophthora species |
| Impact | • Loss of timber value and other public goods and services provided by affected trees |
2
| • Potential for recovery associated with differing disease levels | |
| Management | • Control options for the pathogen in plantation and nursery situations. |
Images of the pest
| Photo 1 | Photo 2 |
| Symptoms of needle browning in the lower crown of western hemlock indicative of P. pluvialis infection Source/ copyright owner: Forest Research | Resin covered canker on 1 cm diameter branch of western hemlock Source/ copyright owner: Forest Research |
3
| Photo 3 | Photo 4 |
| Symptoms of external resin bleeding on hemlock indicative of infection by P. pluvialis Source/ copyright owner: Forest Research | Necrotic lesion in phloem of western hemlock caused by P. pluvialis Source/ copyright owner: Forest Research |
4
Is there a need for a detailed PRA or for a more detailed analysis of particular sections of the PRA? If yes, select the PRA area (UK or EU) and the PRA scheme (UK or EPPO) to be used.
| No | X | ||||
| Yes | PRA area: UK or EU | PRA scheme: UK or EPPO |
Given the information assembled within the time scale required, is statutory action considered appropriate / justified?
[The text below is a recommendation by the risk analyst which requires approval by PHRG]
Yes
Statutory action
No
Statutory action
Phytophthora pluvialis is newly identified in the UK and has already established outdoors in some areas of England, Wales, and Scotland. Due to the developing situation with this pest no final judgement has been made at present as to whether it meets the criteria for a
Quarantine organism. Management options are in place and evidence from all options plus ongoing research findings will continue to be reviewed to update disease management and assess the UK situation.
5
Stage 1: Initiation
1. What is the name of the pest?
Name: Phytophthora pluvialis Reeser, Sutton & Hansen
Synonyms: None
Taxonomy: Kingdom – Chromista: Phyllum – Oomycota; Order – Peronosporales; Family – Peronosporacae; Genus – Phytophthora
Common name: The pest does not have a common name, but the disease that it causes is referred to as red needle cast (RNC) in New Zealand
Etymology: Specific ephithet ‘pluvilalis’ refers to the rain-associated canopy drip in tanwood-Douglas fir forests in western Oregon from which the first
isolates of P. pluvialis were recovered from.
Special notes on taxonomy
The Internal transcribed spacer ITS-based phylogenetic tree for Phytophthora places P. pluvialis in Clade 3a, with closest relatives comprising P. pseudosyringae, P. nemorosa, P. ilicis and P. psychrophila (Abad et al. 2019).
2. What initiated this rapid PRA?
Severe decline was observed in a mature stand of western hemlock (Tsuga heterophylla) in southwest England (UK) in late August 2021, during the Forestry Commission’s annual aerial surveillance for Phytophthora ramorum. Symptomatic material was collected and sent for analysis to the Tree Health Diagnostic and Advisory Service at Forest Research, UK. Analysis of material, including isolation onto Phytophthora-selective medium, yielded a species of Phytophthora. Based on ITS and coxII sequences the species was identified as P. pluvialis (Pérez-Sierra et al. 2022a); identification of P. pluvialis was also confirmed by real-time PCR using the protocol of McDougal et al. (2021). Previously P. pluvialis has only been known from New Zealand and the Pacific Northwest (PNW) states of the USA. It causes minor effects on Douglas fir (Pseudotsuga menziesii) and tanoak (Notholithocarpus densiflorus) in Oregon and wider PNW (Reeser et al. 2013; Hansen et al. 2015) and a more damaging needle disease on radiata pine (Pinus radiata) in New Zealand which Dick et al. (2014) named red needle cast (RNC). The pest was recognised as a potential risk to forestry species in the UK and had been added to the Plant Health Risk Register in 2014, but the recent findings in southwest England require a PRA.
3. What is the PRA area?
The PRA area is the whole of the United Kingdom of Great Britain and Northern Ireland.
6
Stage 2: Risk Assessment
4. What is the pest’s status in the plant health legislation, and in the lists of EPPO1?
The pest is not listed in the EU2 or GB3 plant health legislation and is not recommended for regulation as a quarantine pest by EPPO or on the EPPO Alert List. However, the UK Plant Health Risk Group concluded that P. pluvialis meets the criteria to be classified, at least initially, as a GB quarantine pest for regulatory purposes4.
5. What is the pest’s current geographical distribution?
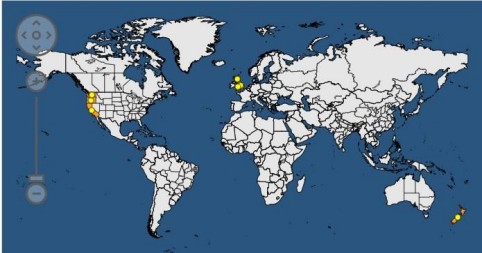
The EPPO Global database shows the current known distribution of the pest (see Figure 1 reproduced from EPPO) with further details in Table 1 below.


1 https://www.eppo.int/ACTIVITIES/quarantine_activities
2 http://data.europa.eu/eli/reg_impl/2019/2072/oj
3 https://www.legislation.gov.uk/uksi/2020/1527/contents/made
7
| Table 1: Distribution of Phytophthora pluvialis (EPPO 2022 and unpublished data) | |
| North America: | Present: California, Oregon, Washington, USA. Extent of the distribution may be under-estimated due to masking of symptoms by other pathogens. |
| Central America: | Absent |
| South America: | Absent |
| Europe: | Present: UK (England, Scotland and Wales) but with restricted distribution. |
| Africa: | Absent |
| Asia: | Absent |
| Oceania: | Present: New Zealand (North and South islands) |
6. Is the pest established or transient, or suspected to be established/transient in the UK/PRA Area?
Yes. Findings of the pest have been made in England, Scotland, and Wales. It has been found infecting established trees in managed forest/woodland environments. There have not been any findings in nurseries. Currently the pest is considered to have a limited distribution and is the subject of official control. Extensive surveillance by the Forestry Commission (FC), Scottish Forestry (SF), Natural Resources Wales (NRW), the Animal & Plant Health Agency (APHA) and Department of Agriculture, Environment and Rural Affairs (DAERA) in Northern Ireland is being undertaken to determine the distribution of P. pluvialis in the PRA area.
Summary of current situation
Between September 2021 and July 2022, P. pluvialis was detected in the wider environment at 13 sites in England (Cornwall, Cumbria, Devon, Shropshire, and Surrey), 18 in Wales (Caerphilly, Carmarthenshire, Conwy, Denbighshire, Gwynedd, Monmouthshire, Rhondda Cynon Taf and Powys) and five in Scotland (Highland, Ross shire and Argyll). At most of these sites western hemlock is the only known host, but both western hemlock and Douglas fir are affected at a few of the commercial forestry plantations. In addition, multiple baits placed in streams in Grizedale Forest (Cumbria) have yielded P. pluvialis although no infected trees have been identified in that forested area There have been no findings in the wider environment in Northern Ireland.
8
7. What are the pest’s natural and experimental host plants; of these, which are of economic and/or environmental importance in the UK/PRA area?
Host plants
All known natural hosts of P. pluvialis are within the family Pinaceae, except for Notholithocarpus densiflorus which is in the Fagaceae. Symptoms incited by P. pluvialis vary with host and are summarised in Table 2, but much of the information is incomplete for minor hosts. Details of symptoms are presented in host and country combinations, as the same disease symptoms are not always apparent in each host for pest outbreaks reported from different countries.
| Table 2: Naturally infected hosts of Phytophthora pluvialis | |||
| Host species | Common name | Disease type | References |
| Notholithocarpus densiflorus | Tanoak | Twig and stem cankers (USA) | Reeser et al. 2013 |
| Larix sp | Larch | Twig cankers (UK) | Records FR THDAS 2022 |
| Pinus radiata | Radiata pine | Needle cast (NZ) | Dick et al. 2014 |
| Pinus patula | Patula pine | Needle cast (NZ) | Scott et al. 2019 |
| Pinus pinea | Stone pine | Needle cast (NZ) | Scion 2022 (unpublished report) |
| Pinus strobus | Weymouth pine | Needle cast (NZ) | Scott et al. 2019 |
| Pseudotsuga mensiezii | Douglas fir | Needle cast (NZ) | Gómez-Gallego et al. 2017 |
| Needle cast and twig lesions (USA) | Hansen et al. 2015 | ||
| Stem cankers and needle cast (UK) | Records FR THDAS 2021; Pérez-Sierra et al. 2022b | ||
| Tsuga heterophylla | Western hemlock | Needle cast (NZ) | Scion 2022 (unpublished report) |
| Twig and stem cankers, needle cast (UK) | Pérez-Sierra et al. 2022a |
New Zealand
Radiata pine and Douglas fir are the primary hosts of P. pluvialis in New Zealand, with single or occasional reports of the agent infecting Pinus pinea (stone pine), P. patula (patula pine), P. strobus (Weymouth pine) and western hemlock. With all hosts, infection
9
appears almost exclusively limited to needles and invariably leads to premature needle loss.
Early symptoms on radiata pine consist of olive-coloured lesions on needles that contain black, resinous bands. The lesions quickly turn khaki-coloured with entire needles then taking on a yellow-brown or red hue before being cast, hence the common name of red needle cast (RNC) coined by Dick et al. (2014). A thick carpet of prematurely shed needles on the forest floor can be a sign of heavy infection. Apart from causing symptoms on needles, P. pluvialis has also been isolated from the rhizosphere of diseased radiata pine grown in bare-rooted nursery beds, but not from naturally infected roots (Scott et al. 2019). However, following artificial inoculation, P. pluvialis has been shown to have the potential to infect and limit the growth of fine roots of radiata pine at apparently sub-lethal levels (Scott et al. 2019).
Symptoms on Douglas fir are similar to those found on radiata pine: needles have olive coloured lesions with black resinous bands, so needles take on an overall mottled, chlorotic appearance (Gómez-Gallego et al. 2017). They are dislodged readily so suitable test material for diagnosis can be lost rapidly (Gardiner et al. 2020). Experimental infection with zoospores of P. pluvialis reproduces the symptoms in Douglas fir, although the presence of another pathogen Nothophaeocryptopus (Phaeocryptopus) gaeumannii (Swiss needle cast) produces similar symptoms and may interact with P. pluvialis or mask its presence (Gómez-Gallego et al. 2017).
For minor hosts occasionally infected by P. pluvialis (patula pine, stone pine, Weymouth pine and western hemlock), there is little detail about the symptoms but banding or necrotic spotting on needles and premature needle loss is mentioned, although the pattern of needle cast differs from that observed on radiata pine (Scion 2022). Disease outbreaks on these minor hosts usually only occur when they are growing adjacent to radiata pine and Douglas fir stands already heavily affected by RNC. This suggests that minor hosts only become infected by P. pluvialis when exposed to high inoculum loads and so are less susceptible to the pathogen.
Pacific Northwest (PNW), USA
Douglas fir and tanoak are the only known hosts of P. pluvialis in the USA. First reports of P. pluvialis came in the absence of any visibly diseased hosts after its recovery from streams, soil samples and canopy drip in mixed tanoak-Douglas fir forest in Oregon (Reeser et al. 2013). Surveys then detected P. pluvialis on rare occasions causing twig and stem cankers on tanoak, although it was found to be only weakly pathogenic when inoculated into tanoak stems (Reeser et al. 2015).
Findings on Douglas fir were made by Hansen et al. (2015) and it is now thought that P. pluvialis is widespread but a usually inconspicuous foliar pathogen when associated with this host (Hansen et al. 2017). Reported symptoms are mainly chlorotic needles that are shed readily. Two-year old seedlings exposed to natural inoculum levels in the forest have been found to develop twig symptoms that included tip dieback and stem lesions extending from bud scars, in addition to premature needle loss and irregular, mottled needle chlorosis. The same type of twig symptoms could not be seen on overstorey trees
10
(Hansen et al. 2015). Similar symptoms have been induced on experimentally inoculated Douglas fir seedlings. However, in winter 2014‐2015, specific symptoms associated with P. pluvialis consisting of dramatic chlorosis/ reddening of needles were observed on Douglas fir trees of all ages in many locations in the central coast range of Oregon. By the following year symptoms had declined and the trees recovered (Hansen et al. 2017).
Great Britain
On the most frequently affected host, western hemlock, P. pluvialis causes dieback, premature needle drops, and branch and stem cankers on both semi-mature and mature trees. Young, naturally regenerated hemlock in the understorey of affected mature trees can also show high levels of dieback and mortality (Pérez-Sierra et al. 2022a). Branch and stem cankers exude copious resin and often have a blackened and cracked surface. When the outer bark of the cankers is removed to expose necrotic lesions in the phloem, resin pockets are usually visible below the lesions and sapwood (Pérez-Sierra et al. 2022a). Just as with radiata pine, it is usually the lower branches of trees that are affected first, with browning needles and multiple cankers on twigs and branches of all sizes. Observations also indicate that cankers can occur at the root collar and on major roots (A. Pérez-Sierra, unpublished data), and fine root systems on understorey hemlock may also be impaired (C.M. Brasier, unpublished data).
With Douglas fir, symptoms are less severe, but include chlorotic and prematurely cast needles, and stem cankers with underlying resin pockets (Pérez-Sierra et al. 2022b). However, as observed elsewhere with this host, N. gaeumannii (Swiss needle cast) infection of needles may also be present and cause similar symptoms to P. pluvialis on needles.
More recently there has been an instance of P. pluvialis infection of larch (Larix sp.), although the circumstances were unusual. Host material was sampled from a low branch submerged in river water, and the pathogen was detected in this water course and also from the sample material. Very possibly, persistent exposure to P. puvialis in the water course may have resulted in infection of the larch tissue contrasting with the aerial infections seen on other hosts.
Apart from satisfying Koch’s Postulates on western hemlock (Pérez-Sierra et al. 2022a), only a small number of other conifer and broadleaf hosts have been challenged with P. pluvialis under controlled conditions in the laboratory to assess the potential host range. Various inoculation methods were used but hosts only developed symptoms if wounded prior to inoculation; summarised findings are shown in Table 3. The response of each host species when inoculated with P. pluvialis was not always consistent, probably due to the genetic differences between P. pluvialis isolates used in the tests which had been selected from the UK, the USA and New Zealand. Inoculation of most hosts resulted in only very limited necrosis around the inoculation point, and holly (Ilex aquifolium), cherry laurel (Prunus laurocerasus), Sitka spruce (Picea sitchensis) and Corsican pine (Pinus nigra subsp. laricio) showed either no or very low susceptibility based on the necrosis that developed. Overall, Douglas fir and western hemlock were the most susceptible hosts, and needles but not the stem bark of radiata pine had some susceptibility. It was noteworthy that the symptoms caused by P. pluvialis were much less severe than those caused by P.
11
ramorum, which was also included in tests of the same tests These preliminary data suggest that that P. pluvialis is a less aggressive pathogen than P. ramorum, another recent introduction to the UK (Webber 2022), and possibly has a more limited host range.
Table 3: Experimental hosts of Phytophthora pluvialis4tested using wound inoculation and outcome of the tests
Host species Common name Plant part tested Susceptibility*
Ornamental plants
| Ilex aquifolium | Holly | Plant stem | None to low |
| Ilex aquifolium | Holly | Detached leaves | None to low |
| Prunus laurocerasus | Cherry laurel | Plant stem | None to low |
| Prunus laurocerasus | Cherry laurel | Detached leaves | Low |
| Rhododendron ponticum var Cunningham’s White | Rhododendron | Plant stem | Low |
| Rhododendron ponticum var Cunningham’s White | Rhododendron | Detached leaves | Low |
| Tree species | |||
| Picea abies | Norway spruce | Plant stem | Low |
| Picea sitchensis | Sitka spruce | Plant stem | None |
| Pinus nigra subsp laricio | Corsican pine | Plant stem | None to low |
| Pinus radiata | Radiata pine | Plant stem | None to low |
| Pinus radiata | Radiata pine | Detached needles | Moderate to high |
| Pinus sylvestris | Scots pine | Plant stem | Low |
| Pseudotsuga menziesii | Douglas fir | Plant stem | Moderate |
| Tsuga heterophylla | Western hemlock | Plant stem | Moderate to high |
During monitoring of water courses using detached sections of foliage or leaves (‘baits’) to detect the presence of P. pluvialis, Douglas fir and western hemlock shoots were colonised by natural available inoculum of the pathogen. Furthermore, on one occasion
4 Data summarised from unpublished 2022 Defra interim report: Investigation on potential susceptible hosts for Phytophthora pluvialis in forests, wider environment and horticulture. Authors: FERA: A. Barnes, A. Elliott, C. Field, A. Ozolina & L. Elliott; FR: M. Crampton, A. Eacock, : R. Chitty, C. Gorton & .A. Pérez-Sierra; SASA: A. Schlenzig, L. Feehan & R. Campbell
12
foliage of Koyama spruce (Picea koyamae) and very occasionally foliage of Sitka spruce (P. sitchensis) acted as a bait for P. pluvialis possibly suggesting some susceptibility, although this was not confirmed by the limited host testing shown in Table 3.
Economic and environmental importance of host plants
Tanoak is occasionally planted in the UK as an ornamental tree, with very few suppliers of the species listed in Britain5. Radiata pine is more common, but forestry plantings are mainly limited to provenance trials (see https://www.forestresearch.gov.uk/tools-and resources/tree-species-database/radiata-pine-monterey-pine-rap/). The Botanical Society of Britain and Ireland (BSBI) Atlas (Figure 2) shows the distribution of the species as mainly limited to southwest England and the coastal regions of Wales and Southern England, with very sporadic records in the central belt right up to Northeast Scotland. Douglas fir is considered a principal tree species, widely used for forestry and a popular and proven option to diversify production forests in line with the UK forestry standard6. It is found throughout much of the British Isles (Figure 2). Planted areas comprise just under 60,000 ha across Britain (Table 4), and amount to about 2% of the total conifer stock in Britain7. In contrast, western hemlock is considered a minor forestry species (less than 0.5% of the total conifer stock) and has a more limited distribution (Table 4) with much of the 9,000 ha of stock concentrated in southern and southwest England (Figure 2; Harmer et al. 2011). None of the known hosts are native to Britain which reduces their value to the environment.
| Table 4: Planted areas of Douglas fir and western hemlock in Britain (unpublished data from National Forest Inventory database, 2022) | ||||
| Conifer species | England | Scotland | Wales | Great Britain |
| Douglas fir | 24,751 ha | 24,565 ha | 9,690 ha | 59,006 ha |
| Western hemlock | 5,706 ha | 1,849 ha | 1,512 ha | 9,067 ha |
| Total | 30,457 ha | 26,414 ha | 11,202 ha | 68,073 ha |
5 RHS Plantfinder https://www.rhs.org.uk/plants/nurseries-search-result?query=10348 6 https://www.gov.uk/government/publications/the-uk-forestry-standard
7 Forestry Statistics 2021: https://www.forestresearch.gov.uk/tools-and-resources/statistics/data-downloads/
13



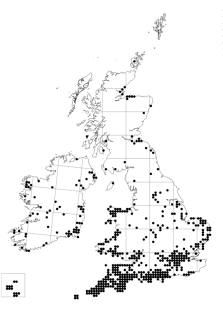

Radiata pine Douglas fir Western hemlockRadiata pine Douglas fir Western hemlockRadiata pine Douglas fir Western hemlock
Figure 2: Distribution of radiata pine, Douglas fir and western hemlock based on Botanical Society of Britain and Ireland (BSBI) maps showing presence in 10 km squares in Great Britain and Ireland https://bsbi.org/maps
14
8. Summary of pest biology and/or lifecycle
Phytophthora pluvialis is a homothallic species, meaning it can undergo sexual reproduction and as a result produce oogonia (sexual spores) in single-strain culture. Phytophthora oogonia are often considered to be persistent spores, able to survive for weeks or months under suboptimal conditions (Erwin & Ribeiro 1996; Boevink et al. 2020). The more ephemeral spores, free-swimming zoospores which are initially contained within sporangia, are formed through vegetative (asexual) reproduction. The sporangia are semi papillate and partially caducous (easily shed) (Reeser et al. 2013) indicating their potential for aerial dispersal, in rain splash and fog.
Phytophthora pluvialis causes a polycyclic disease, and studies of the pathogen in New Zealand have shown there are several infection cycles per year that are closely associated with rainfall (Gómez-Gallego et al. 2019a; Williams & Hansen 2018). Spores (presumed to mostly be sporangia/ zoospores) of P. pluvialis are released from infected foliage during the cooler period of the year between autumn and spring (Fraser et al. 2020). If free-water is present on needle surfaces infection by spores can occur within 18 hours, with proliferation in plant tissue producing masses of sporangia which start to protrude from needle stomata a few days later (Gómez-Gallego et al. 2019a). Providing there are conducive weather conditions, reinfection cycles will occur every 4-6 days. In New Zealand the first symptoms of RNC usually appear in autumn or winter on the lower branches of affected trees, but under favourable conditions the disease can spread up the crown and to neighbouring trees. Where symptoms are limited to the lower branches, and sporulation occurs on infected foliage on these branches, aerial dispersal of P. pluvialis may remain largely within the understorey layer and the potential for longer distance dispersal above the tree canopy be much more limited.
Infected needles are cast by early to mid-spring in the following year (Dick et al. 2014; Fraser et al. 2020). It is unclear once the needles are cast and in the litter layer, how long the inoculum they contain can persist in viable form and if it plays a further role in the disease cycle. To understand this aspect of the disease cycle, the longevity of P. pluvialis in the litter layer exposed to UK conditions is currently under investigation. Potentially, inoculum of oospores and asexual resting structures (e.g. encysted zoospores, stromata, hyphal swellings/ aggregations) of P. pluvialis could be present in the litter layer and rhizosphere, and act as a reservoir of genetic diversity and inoculum for reinfection. Typically, oospores are considered resistant to extreme environmental conditions, allowing them to survive for several years in soil or host plant material (Erwin & Ribeiro 1996). However, oospores or other resilient structures have rarely been observed in plant material (Hood et al. 2014; Williams & Hansen 2018) so the mode of survival by P. pluvialis through warmer/drier summer months has yet to be elucidated. Under favourable conditions for P. pluvialis, affected trees that were completely green at the start of autumn can be defoliated almost completely by the following spring, but the new growth is seldom affected (Dick et al. 2014). Therefore, the disease affects photosynthetic capability and hence growth, but does not cause tree mortality at least in New Zealand (Ganley et al. 2014). In the USA, P. pluvialis is considered an inconspicuous foliar pathogen which is
15
probably native and causes relatively little damage to hosts (Reeser et al. 2013; Hansen et al. 2017).
9. What pathways provide opportunities for the pest to enter and transfer to a suitable host and what is the likelihood of entering the UK/PRA area?
Phytophthora pluvialis has been confirmed at 36 sites (England, Scotland, and Wales) across the PRA area. The most northerly finding is in Ross-shire8, furthest west in Cornwall, and most easterly in Surrey. In the same way that it is now considered that P. pluvialis was introduced into New Zealand several years before RNC became noticeable on radiata pine in 2008 (Dick et al. 2014), it is also likely that the pathogen has been in the UK for some time although visible signs of disease may only have emerged in 2021, possibly in response to a sequence of years with favourable weather conditions (Defra, unpublished data 2022). For many of the affected sites, symptoms observed in autumn through to spring in 2021-22 may not have been noticed or considered concerning were it not for the specific surveys and interest triggered by the finding of P. pluvialis on some heavily affected trees at a site in the southwest of England. Several possible pathways exist for entry into regions of the PRA area, with some likely to be direct/principal pathways (plants, wood) and others probably less significant or indirect pathways (seeds, foliage, soil, and machinery). The risk from each pathway is assessed below.
Plants for planting (trade) pathway
Various studies have reported the frequency with which Phytophthoras are associated with plants in nurseries and how such infected plants then act as a pathway to introduce these pathogens into natural, semi-natural and horticultural ecosystems (e.g., Jung et al. 2016). In the context of P. pluvialis current evidence suggests the most likely pathway for introduction is on live conifer plants, particularly Douglas fir and radiata pine, although potentially other Pinus species could be hosts. In Great Britain, however, the Plant Health (Phytosanitary Conditions) (Amendment) (EU Exit) Regulations 2020, Annex 6, Part A (1.) prohibits the import of conifer plant species (Abies, Cedrus, Chamaecyparis, Juniperus, Larix, Picea, Pinus Pseudotsuga and Tsuga) other than as seeds, from all third countries other than EU member states and other specified European countries9. Identical requirements apply in Northern Ireland, under Commission Implementing Regulation (EU) 2019/2072 (Annex VI, item (1). Although the introduction of P. pluvialis into the UK probably pre-dates the most recent regulation, prohibition of conifer species from third countries has been in place for many years in the form of much earlier legislation applied to the Import and Export of Trees, Wood and Bark (Health) (Great Britain) Order 1980 and
8 https://forestry.gov.scot/sustainable-forestry/tree-health/tree-pests-and-diseases/phytophthora-pluvialis 9 Specified European countries and areas are Albania, Andorra, Armenia, Azerbaijan, Belarus, Bosnia and Herzegovina, Canary Islands, Faroe Islands, Georgia, Iceland, Liechtenstein, Moldova, Monaco, Montenegro, North Macedonia, Norway, specific parts of Russia, San Marino, Serbia, Switzerland, Turkey and Ukraine.
16
the Tree Pests (Great Britain) Order 1980. On this basis, entry via traded conifer plants for planting from North America (Canada, USA Pacific Northwest) and New Zealand where P. pluvialis is known to occur, is very unlikely. Likewise, plant health surveillance programmes across Europe have not made any detections of P. pluvialis in nurseries or the wider environment. Despite this, there are uncertainties associated with the plants for planting pathway such as the lack of information on the wider host range of P. pluvialis, especially in relation to broadleaf hosts, although the limited host testing undertaken so far has indicated that other conifers such as Scots and Corsican pine, and Sitka spruce show either no or low susceptibility (Table 3). Additionally, although Tabima et al. (2021) suggest that P. pluvialis is likely to have originated in Oregon’s coastal Douglas fir forests of the Pacific Northwest (PNW), with at least one introduction event from the PNW allowing it to establish in New Zealand, they also comment that “the potential exists for undiscovered populations of P. pluvialis” beyond the PNW and New Zealand.
Plants for planting (trade).
Pathway 1 Very
unlikely Unlikely Moderately
| ✓ |
likely Likely Very
likely
Confidence High
Medium
Low
| ✓ |
Confidence
Confidence
Confidence
Overall, therefore, the plants for planting pathway is rated as very unlikely based on current and earlier import regulations applied to conifers although with low confidence because of uncertainties about host range, particularly broadleaf species, and the wider geographical distribution of P. pluvialis.
17
Plants for planting (non-trade) pathway
Due to the mild climate of southwest England and indeed much of western Britain, this region is rich in gardens open to the public which specialise in unusual or novel specimen ornamental shrubs and trees collected from many parts of the world10. Such plant collecting activities undertaken by both professionals and amateurs could potentially be a non-trade plant pathway for entry. Until 5-10 years ago, the biosecurity risks posed by these activities were rarely considered and collected plants brought in by this pathway were seldom quarantined or monitored after planting out to mitigate any risks of accidental pest introduction (Webber 2010). However, in comparison to the trade plant pathway, this non-trade plant pathway is relatively minor and sporadic, although likely to encompass a wide range of plant genera some of which may be currently unknown hosts or non-traded hosts of P. pluvialis (e.g. tanoak). Considering the size of this pathway and the lack of awareness about associated biosecurity risks, it is rated as unlikely although with low confidence due to lack of data about plant numbers and uncertainty about the host range and geographical distribution of P. pluvialis.
Plants for planting (non-trade).
Pathway 2Very
unlikely Unlikely Moderately
likely Likely Very
likely
Confidence High
Medium
| ✓ |
Low
| ✓ |
Confidence
Timber/Wood pathway
Confidence
Confidence
Due to the export market for radiata pine grown in New Zealand, Hood et al. (2014) explored in depth the likelihood of movement of P. pluvialis on pine logs in the form of viable spores on bark surfaces or as colonised bark or sapwood. They concluded, based on loss of spore viability when tested as inoculum directly or in infected pine needles, coupled with the inability of P. pluvialis to colonise bark or sapwood of radiata pine, that neither logs or sawn timber were a likely pathway for the transport of the pathogen. Similar studies have not been undertaken with needles of Douglas fir, but stem lesion/canker formation has not been seen for this host in either New Zealand or the USA and spore viability is likely to be the same (Dick et al. 2014; Hansen et al. 2015).
In contrast, cankers have been found to form on the stems/trunks of both hemlock and Douglas fir in the UK, with P. pluvialis invading phloem and cambial tissues and penetrating the sapwood. An assessment of 97 cankers on trunks of western hemlock and Douglas fir trees infected by P. pluvialis indicated that sapwood penetration was mostly
10 Examples of plant collection gardens can be found at https://www.nationaltrust.org.uk/lists/top-gardens to-visit-in-the-south-west
18
superficial and usually limited to 2-5 mm (J.F. Webber et al, 2022). Where deeper penetration into the sapwood was occasionally detected, it was in fewer than 25% of cankers on western hemlock only 5% associated with Douglas fir. The maximum depth P. pluvialis detected in Douglas fir sapwood was 9 mm and 15 mm for hemlock, but in both species this depth only applied to a single canker (Webber et al. 2022). However, any conifer wood or wood packaging material (WPM) to be imported into GB or Northern Ireland from EU or third countries including the USA and New Zealand must meet at least one of three phytosanitary requirements: (i) to be bark-free, (ii) to come from an area free from specified bark beetle pests or (iii) to be heat treated (56oC for 30 minutes). Thus, even if P. pluvialis cankers are more widespread on conifer stems than is currently known, removal of the bark would ensure elimination of most infected material. Hood et al. (2014) also found that P. pluvialis oospores were unlikely to be viable after exposure for 2 hr at 35°C, suggesting required wood treatment at higher temperatures of 56oC for 30 minutes would destroy any residual traces of P. pluvialis including any mycelium or oospores in sapwood.
Timber
Pathway 3Very
unlikely Unlikely Moderately
| ✓ |
likely Likely Very
likely
Confidence High
| ✓ |
Medium
Low
Confidence
Confidence
Confidence
The likelihood of entry via logs/wood/WPM is therefore assessed as very unlikely and with high confidence due to the poor ability of the pest to survive as a hitchhiker (possibly in the form of infected needles/needle fragments) on the exterior of wood, the lack of stem lesions on radiata pine or Douglas fir which infer that the pathogen is not present in bark or sapwood, and the phytosanitary treatments required for imported conifer wood or WPM which would remove P. puvialis if present in bark and also in sapwood if heat treatment was applied. Stem cankers have only been recorded on tanoak on rare occasions (Reeser et al. 2013) and it is not a timber species, so again it is very unlikely that timber from this host would act as a pathway.
Cut Foliage pathway
Typically, foliage is collected from the wild for ornamental uses, it often comes from conifers, and is not subject to any specific cultural or treatment practices other than perhaps drying. As already described in Section 8, P. pluvialis has been found to produce abundant sporangia on infected needles of radiata pine (Gómez-Gallego et al. 2019a;) and by extension on infected needles of Douglas fir (Gómez-Gallego et al. 2019b). Therefore, dried foliage or cut branches from susceptible hosts may contain viable pathogen structures, especially if formed within infected tissues, although drying (especially if using heat) is likely to reduce pathogen viability in or on the plant tissue. Material may also be dyed, bleached, or otherwise impregnated which may also reduce pathogen prevalence.
However, just as with plants for planting, the import of conifer foliage from all third countries is prohibited under GB Plant Health Regulations (see above). Additionally,
19
oospores (likely to be the most resilient spore structures of P. pluvialis) have rarely been observed in infected needles of radiata pine (Hood et al. 2014; Williams & Hansen 2018), suggesting that survival by P. pluvialis during transport on this pathway is likely to be short-lived, especially if foliage is dried or subject to other treatments.
Therefore, the likelihood of entry via cut foliage is assessed as very unlikely although as cut foliage might also include hosts other than the currently known hosts, or Douglas fir for which there is a lack of information about the presence of oospores in infected needles, medium confidence is placed on this pathway rating.
Cut foliage
Pathway 4Very
unlikely Unlikely Moderately
| ✓ |
likely Likely Very
likely
Confidence High
Medium
| ✓ |
Low
Confidence
Seed pathway
Confidence
Confidence
In general, Phytophthora species are not considered to be seed borne pathogens. However, some examples exist: Phytophthora cactorum can be carried on beechnuts of Fagus (Prochazkova & Jancarek 1991), and a recent study demonstrated that seed infection by Phytophthora gemini of a common seagrass (Zostera marina) occurred frequently (Glovers et al. 2016). Even if seed is not infected directly, fruits or cones may become contaminated with soil, or be mixed with debris such as needles or other plant host material that contains pathogen propagules. Particularly with pathogens that infect needles, such as P. pluvialis, there is potential for conifer seed associated with infected debris to act as a pathway. Although plant health regulations prohibit the import of plants of conifer species (see above), seeds from North America and elsewhere are exempt from this measure. Since 1920, hundreds of kilos of seed collected from Douglas fir, western hemlock and radiata pine in North America have been imported into the UK (Anon 1965). More recently, data on forestry imports of conifer seed (Table 5) suggests that Douglas fir seed is still regularly imported into the UK from North America, so this pathway remains for this species at least.
| Table 5: Quantity (in kg) of conifer seed imported into the UK from the USA and Canada since 2004 (for years not shown in table no seed imports were recorded) (Forestry Commission Plant Health (2022) | |||||||||||||
| 2004 | 2005 | 2008 | 2009 | 2010 | 2012 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | |
| Douglas fir | 5 | 18 | 186 | 67 | 72 | 18 | 133 | 16 | 23 | 67 | 6 | 17 | 29 |
| Western hemlock | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.5 | 0 |
| Radiata pine | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
20
However, if few or no long-lived spore stages, such as oospores, are produced in needles by P. pluvialis (Hood et al. 2014; Williams & Hansen 2018) this would limit the ability to persist in contaminated soil/needle debris associated with seeds. For these reasons this pathway is assessed as unlikely but with low confidence in the rating due to lack of data of oospore formation in infected foliage of Douglas fir and the extent of debris contamination in imported seed lots.
Seed
Pathway 5Very
unlikely Unlikely Moderately
likely Likely Very
likely
Confidence High
Medium
| ✓ |
Low
| ✓ |
Confidence
Confidence
Confidence
Machinery and soil pathway
In speculating about the transfer of P. pluvialis from the coast range forests of Oregon to New Zealand, Tabima et al. (2021) suggest it could have occurred through various modes of introduction from infected Douglas fir needles to infested soil, due to lax biosecurity practices. In this context, Brar et al. (2017) mention that imported machinery could have been a possible pathway for P. pluvialis, as prior to 2001 the risks associated with used forestry machinery were not recognised in New Zealand (or probably elsewhere). As already described in Section 8, P. pluvialis has been found to produce abundant sporangia on infected needles of radiata pine (Gomez-Gallego et al. 2019a) and by extension on infected needles of Douglas fir (Gomez-Gallego et al. 2019a). As these needles die and fall to the forest floor often forming a thick carpet of prematurely shed needles, it creates potential for the pathogen to be harboured in soil and for contaminated soil to act as a pathway. However, as already indicated above, P. pluvialis may not persist for long in viable form in fallen needles and soil due to the lack of long-lived spore stages (Hood et al. 2014; Williams & Hansen 2018). In addition, importation of soil from third countries such as the USA and New Zealand is already prohibited under the Plant Health (Phytosanitary Conditions) (Amendment) (EU Exit) Regulations 2020 for Great Britain, and Commission Implementing Regulation (EU) 2019/2072 for Northern Ireland. However, machinery and vehicles which have been operated for forestry purposes can be imported providing they are accompanied by a phytosanitary certificate, and this should ensure that any risk is mitigated. Based on current regulation, this pathway is assessed as very unlikely, but with medium confidence due to the lack of data on how frequently used forestry machinery is imported into the UK from countries where P. pluvialis is present and levels of compliance to make sure there is no associated soil/forestry debris with such machinery.
Machinery and soil
21
Pathway 6Very
unlikely Unlikely Moderately
| ✓ |
likely Likely Very
likely
Confidence High
Medium
| ✓ |
Low
Confidence
Confidence
Confidence
10. If the pest needs a vector, is it present in the UK/PRA area?
Phytophthora pluvialis does not require a vector for dispersal.
11. How likely is the pest to establish outdoors or under protection in the UK/PRA area?
Under protection
Establishment under protection is assessed as very unlikely with high confidence, because hosts of P. pluvialis are not grown under protection for their life span.
Under
unlikely Unlikely Moderately
Very
| ✓ |
likely Likely Very
Protection
Confidence High Confidence
Outdoors
| ✓ |
Medium
Confidence
Low
Confidence
likely
Host susceptibility and distribution influence the likelihood of P. pluvialis establishment in the wider environment; with climatic factors, such as temperature, moisture and needle wetness also playing a part in how likely outbreaks are to establish and the intensity at which they occur. Suitable hosts are also present in the UK (section 7; Figure 2).
In culture, P. pluvialis grows readily at between 15-20oC, with no growth above 25oC (Reeser et al. 2013), which is consistent with a lack of detections of P. pluvialis above a mean maximum temperature of 21.6oC in New Zealand (Fraser et al. 2020). Rainfall and relative humidity (RH) have also been highlighted as important drivers of RNC disease development and sporulation on radiata pine by Fraser et al. (2020). They found that sporulation and infection take place in the coolest, wettest part of the year (mid-winter through to mid-spring) peaking in June-August in New Zealand. At this time of year rainfall and RH are at their highest and average air temperatures at their lowest (Table 6; Scion 2022).
22
Table 6: Monthly averages for 2015-2020 in New Zealand* (Scion 2022)
Month
Average Air Temp. (°C)
Average RH (%)
Average Total Rainfall (mm)
Jan 17.01 79.33 93.9
Feb 16.70 82.18 116.5
Mar 14.72 86.72 201.7
Apr 12.32 86.38 216.5
May 10.49 84.82 180.4
Jun 8.41 88.07 286.7
Jul 7.24 86.94 251.9
Aug 7.66 85.87 151.9
Sep 8.95 83.59 292.5
Oct 11.35 80.23 141.2
Nov 13.31 79.88 132.2
Dec 14.64 81.44 174.5
Grand Average 11.89 83.79 186.7
* Means for selected sites higher elevation coastal sites on the east coast of the North Island
Mild, wet winters, particularly those that are not too cold, are most likely to encourage the disease to establish, and in New Zealand it has been found that the most consistent disease expression is on higher elevation coastal sites (greater than 500m such as plateaus or ridge tops) on the east coast of the North Island with favourable relative humidity and rainfall (Scion 2022). The experience in the PNW with disease expression of P. pluvialis on Douglas fir is that it operates under similar climatic factors, except that the colder winters of the PNW tend to limit infections, as does the lower relative humidity in spring (Gómez-Gallego et al. 2019b) so overall levels of disease tend to be much lower compared with New Zealand and are also consistent with a native pathogen co-evolved with a native host.
In New Zealand and the cascades region of Oregon where P. pluvialis is active, the climate is probably very similar to western Britain. Indeed, many of the key commercial forestry conifers grown in Britain (Sitka spruce – Picea sitchensis, lodgepole pine – Pinus contorta, Douglas fir and western hemlock) have been chosen from the PNW because the climate similarities with Britain allow these timber producing species to thrive. Analysis of daily weather variables (temperature and rainfall) at a spatial resolution of 1km and using data available from 2001-2020, has visualised the parts of the UK where the risk of P. pluvialis establishment is likely to be greatest (Figure 3). The risk map shown below emphasises the suitability of climatic conditions throughout western Britain for P. pluvialis (Fera 2022). Additionally, the known hosts of P. pluvialis (Douglas fir, western hemlock and radiata pine) are most numerous in southwest England and Wales (Figure 2), where some of the most favourable climatic conditions occur. Between September 2021 and March 2022, findings of P. pluvialis in the UK were made almost exclusively in western Britain, with locations ranging from the far north of Western Scotland (Ross-shire) down to southwest England (Cornwall). In addition, a single finding was made in Surrey where the
23
climate is likely to less conducive, suggesting that establishment can occur even beyond the areas predicted to be most favourable for P. pluvialis.
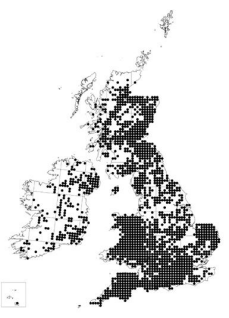
Figure 3: Risk Map based on number of rainy days and temperature indicating areas likely to be most suitable for the establishment of P. pluvialis.
24
Overall, this confirms that a suitable climate for the pathogen is present through much of the PRA area. Similar climate conditions in the North Island of New Zealand, the PNW and particularly the western side of the UK, already favour other introduced aerial Phytophthora species such as P. ramorum (UK and PNW) and P. kernoviae (UK and NZ) and have allowed them to establish. Therefore, environmental conditions throughout at least western UK are predicted to be favourable to P. pluvialis so establishment is very likely and indeed has occurred already at some locations. On that basis the likelihood of establishment in the UK/PRA area is assessed as very likely with high confidence.
Outdoors Very
unlikely Unlikely Moderately
likely Likely Very
| ✓ |
likely
Confidence High
| ✓ |
Medium
Low
Confidence
Confidence
Confidence
12. How quickly could the pest spread in the UK/PRA area?
Speed of spread will be influenced by several factors: the number of introductions and their spatial distribution, the number and distribution of suitable host plants, behaviour of the pest, and fluctuations in environmental conditions that influence pest behaviour and dispersal.
Natural spread
Although the biology of P. pluvialis is not fully understood, it is known that natural spread occurs via partially caducous sporangia that are produced on infected needles in the tree canopy (Dick et al. 2014; Williams & Hansen 2018). These easily detached sporangia are
adapted for aerial dispersal in rain splash and fog. In New Zealand there has been no systematic work on the dispersal distances of sporangia, but most P. pluvialis spread is probably over short distances as illustrated by the localised disease expression within trees and forests. However, since RNC was first formally identified in New Zealand in 2008, findings of P. pluvialis have increased markedly (Graham et al. 2018). This, plus genotyping evidence that points to a single introduction followed by clonal spread (Tabima et al. 2021), suggests that spread by P. pluvialis has been moderately rapid over one to two decades. In addition, there are records of isolated trees and windbreaks that have become infected although some distance from another outbreak, indicating that longer distance aerial dispersal ‘jumps’ are also possible (Scion, 2022).
Apart from the arial dispersal of inoculum by fog and wind-driven rain, natural spread of P. pluvialis will also aided by movement of spores in water courses and run-off water, a common occurrence with most Phytophthoras. Leaf baiting methods to detect P. pluvialis in water courses in Britain (see Section 7), have mainly been aimed at monitoring sporulation events, but inoculum has been detected at least 500 m downstream from areas with heavily diseased trees (Ana Pérez-Sierra, unpublished data). However, dilution
25
effects as inoculum is moved away from disease foci in water courses probably limits pathogen spread through this process to relatively short distances (e.g. a few hundred metres), especially as the spore stages (sporangia and zoospores) are likely to be relatively short-lived and ephemeral.
Natural
slowly Slowly Moderate
Very
paceQuickly Very
Spread
Confidence High Confidence
Medium
Confidence
| ✓ |
Low
Confidence
| ✓ |
quickly
On that basis, spread is considered likely to occur at a moderate pace, but this will be influenced by climate, the range and spatial distribution of hosts species, and possibly the influence of water courses around disease foci. Due to the lack of information on aerial dispersal distances and water course dispersal, confidence in this rating is placed at medium.
Spread via trade
A major pathway by which P. pluvialis is likely to spread (by analogy with other Phytophthora spp.) is on ‘plants for planting’ of known natural hosts but other host material such as timber and cut foliage may also sustain spread.
As only limited information is available on the full host range, there is also potential for spread on other species of Pinus as well as non-Pinus hosts. Currently there is no evidence to suggest P. pluvialis is circulating in the plant trade, despite the scattered outbreak findings in the UK (Section 6 – Summary). However, movement of infected plants in trade would allow long-distance jumps and new disease foci to be initiated so spread could occur quickly or even very quickly. Although there is a lack of information on the extent of the host range, the testing conducted so far (see Table 3) suggests that P. pluvialis has a limited host range and major UK conifer species such as Sitka spruce, Scots pine and Corsican pine have little or no susceptibility to bark infections. However, the initiating source of the pathogen causing the current outbreaks in the UK is unknown, hence medium confidence in this rating.
Plants for planting
Pathway 1 Very
slowly Slowly Moderate
paceQuickly Very
quickly
Confidence High
Medium
| ✓ |
Low
| ✓ |
Confidence
Confidence
Confidence
Traded forest products will also take the form of logs or timber. When Hood et al. (2014) assessed the likelihood of movement of P. pluvialis on radiata pine logs in the export trade from New Zealand, they concluded the risk was minimal but the assessment was based on infected needles as the only source of inoculum and the inability of P. pluvialis to colonise
26
the bark or sapwood of radiata pine. This contrasts with the disease caused by P. pluvialis in Britain, in which not only the needles of western hemlock and Douglas fir are infected, but also bark in the form of cankers on twigs, branches and main stems of both host species (Pérez-Sierra et al. 2022a, b). The cankers which form on the main stem of both hosts can be extensive, with P. pluvialis invading phloem and cambial tissues and penetrating into the sapwood, although sapwood penetration appears to mostly superficial (see Section 9; Webber et al. 2022) it potentially provides another opportunity for spread.
The association of P. pluvialis with logs/timber may be as a hitchhiker (possibly in the form of infected needles/needle fragments) on the exterior of logs (although if the fragments are oospore-free then viability of P. pluvialis is likely to be short-lived, see Section 8), or in infected phloem and sapwood underlying stem cankers. In addition, the ability of P. pluvialis to sporulate in canker tissues is also likely to influence the likelihood of disease spread via infected logs/timber. Current evidence suggests that sporulation by P. pluvialis in the infected phloem of cankers is likely to be limited but occur mainly or entirely on leaves, needles and shoots where the sterols that are required for sporulation by phytophthoras are most abundant and the inhibitory compounds which prevent sterol uptake such as tannins and phenolics less concentrated (Erwin & Ribeiro 1996; Strong et al. 2013; Wang et al. 2021). Limitations on sporulation in or on timber or logs will inevitably reduce the potential for spread to new hosts. A suitable environment will also be required to encourage sporulation. Additionally, if log material harvested from P. pluvialis affected sites is subject to biosecurity precautions that are already in place for P. ramorum11, aimed at minimising spread during handling and movement from forests and at the processing mill as well as excluding potentially infected material from the woodchain, this would also reduce the risk of P. puvialis spread. Overall spread via timber or logs will is likely to occur slowly but the uncertainty in relation to the viability of inoculum and quantity of sporulation reduces confidence in this assessment to medium.
Timber/logs
Pathway 2 Very
slowly Slowly Moderate
| ✓ |
paceQuickly Very
quickly
Confidence High
Medium
| ✓ |
Low
Confidence
Confidence
Confidence
Foliage for ornamental uses (e.g. wreaths, flower arrangements) can be traded and may contain viable pathogen structures within infected tissues of susceptible hosts, although treatment of the foliage such as drying, dying, or bleaching is likely to reduce pathogen viability. After ornamental use, however, cut foliage is usually discarded into general waste which would limit potential for pathogen spread to susceptible hosts. On that basis, spread
11 Phytophthora manual 9: https://www.forestresearch.gov.uk/tools-and-resources/fthr/pest-and-disease resources/ramorum-disease-phytophthora-ramorum/phytophthora-manual-9-licences-to-move-and-process wood-from-trees-with-ramorum-disease/
27
through traded foliage is considered likely to occur only very slowly with high confidence in this assessment.
Cut foliage
Pathway 3 Very
slowly Slowly Moderate
| ✓ |
paceQuickly Very
quickly
Confidence High
| ✓ |
Medium
Low
Confidence
Confidence
Confidence
13. What is the pest’s economic, environmental, and social impact within its existing distribution?
Douglas fir is considered one of the most important forest trees in Oregon (USA) and the second most valuable commercial conifer species for forestry in New Zealand; radiata pine is the most valuable conifer species in New Zealand. In comparisons of Douglas fir (New Zealand vs USA) evaluations indicate that P. pluvialis is much more abundant in New Zealand but causes much less conspicuous damage in the PNW (Gómez-Gallego et al. 2019b).
Reeser et al. (2015) suggest that P. pluvialis is “part of the native forest mycota of western Oregon”, and its association with lower canopy needle loss in dense stands located in humid areas may be part of natural turnover, although P. pluvialis may also reduce the success of natural regeneration in dense Douglas fir stands. In New Zealand, however, the impact on plantation radiata pine and Douglas fir is of concern to forest growers in disease prone areas due to losses in increment growth that follows serious episodes of needle cast. Timing of disease expression and severity can also differ markedly between regions and years due to the influence of climate which makes quantification of increment losses associated with RNC difficult (Ganley et al. 2014). In an area where RNC was severe, the annual incremental growth of radiata pine decreased by ~35% in the following year, but growth losses do not appear to persist unless there is repeated defoliation. Williams & Hansen (2018) also comment that in some areas or years the incidence of RNC is so low that it is unlikely to result in any significant reductions in tree growth. Tree mortality due to P. pluvialis infection has never been observed in either Oregon or New Zealand on any host, but it cannot be ruled out that it may predispose trees to attack by secondary pests and pathogens (Ganley et al. 2014).
Based largely on the NZ experience with RNC where damage can be significant but intermittent, the impacts (economic/environmental/social) are rated as medium but with low confidence. There are major difficulties in rating the impact of a likely native pathogen co-evolved with a native host (Douglas fir) for which impacts are minor in the PNW, in combination with the same host and pathogen, but in the latter case where the pathogen has been introduced into a highly managed environment of plantation grown exotic tree species with differing environmental and social values (New Zealand). There is also a lack of quantitative data from both the PNW and NZ to make the assessment.
28
Impacts Very
small Small Medium Large Very
large
Confidence High
Medium
Low
| ✓ |
| ✓ |
Confidence
Confidence
Confidence
14. What is the pest’s potential to cause economic, environmental, and social impacts in the UK/PRA area?
None of the known hosts of P. pluvialis are British native species, and the three most significantly affected (radiata pine, Douglas fir and western hemlock), are non-native species that originate from North America. The resource they provide is estimated at
approximately 68,000 ha of forest cover in Britain, with an annual value of over £80 million, set within the context of a total of 1,308,000 ha of conifer cover in Britain. Only Douglas fir is considered a major forestry species of the known host species (see Section 7). Collectively, however, all pine species grown in Britain make up a much larger proportion of the plantation stock and if Scots or lodgepole pine proved to be susceptible, this would put a much larger number of trees and forest cover at risk (see Table 7 below).
| Table 7: Areas (000 ha) and value of Potential Phytophthora pluvialis hosts in Great Britain shown by country (unpublished data from Defra and National Forest Inventory database, 2022) | |||||
| Principal Species | England | Wales | Scotland | GB | Annual value of GB Woodlands (ONS GB Value) |
| Scots pine (Pinus sylvestris) | 57 | 4 | 138 | 199 | £237,052,017 |
| Corsican pine (Pinus laricicola) | 36 | 2 | 3 | 40* | £47,928,876 |
| Lodgepole pine (Pinus contorta) | 6 | 4 | 75 | 85* | £101,373,972 |
| Douglas fir (Pseudotsuga menziesii) | 25 | 9 | 25 | 59 | £70,361,030 |
| Western hemlock | 6 | 1 | 2 | 9 | £10,811,840 |
| Other pines | 28 | 4 | 31 | 63 | £74,887,527 |
| Total annual value | £542,415,262 |
* Totals may reflect rounding
Potential economic impacts in the PRA area
Economic impacts would not only depend on the number of tree species that might be susceptible to P. pluvialis infection, but also how severe disease symptoms could be and what parts of trees are affected. In the PNW and New Zealand, symptoms are limited to foliage, so hosts affected by severe episodes of P. pluvialis infection have temporarily reduced annual growth increment but apparently recover and are not killed by the pathogen. In contrast, P. pluvialis infection of western hemlock in the UK appears much more damaging, causing dieback and some mortality (unquantified), with cankers
29
developing on the main tree stem that could affect the timber value at harvest. Symptoms are less damaging on Douglas fir, but growth and therefore productivity is likely to be affected and stem cankers may again affect timber values. Currently P. pluvialis is being treated as a regulated pest, and tree felling is required with associated costs. The potential for timber salvage from the infected trees once felled is under investigation and will depend on the extent of sapwood penetration by P. pluvialis if infected wood has to be removed from the supply chain.
On that basis, economic impacts have the potential to be large, particularly if new/additional host species are found to be susceptible and become diseased. Annual value of conifer species at risk is estimated at over £500 million (Table 6), but uncertainty about the host range, the potential of affected trees to recover from infection episodes (some evidence is emerging to suggest this is happening) and whether it will be a continuing requirement to fell affected trees (plus any trees in a specified buffer zone) all affect the rating given for economic impacts. This is reflected in the low confidence rating applied to the assessment.
small Small Medium Large Very
Economic Impacts
Very
| ✓ |
large
Confidence High Confidence
Medium
Confidence
Low
Confidence
| ✓ |
Potential environmental impacts in the PRA area
Environmental impacts include a range of ecosystem services. Thus, removal of large numbers of trees which might be required to deal with outbreaks could result in changes to soil (NH4, NO2 and cations), and on some sites soil erosion, changes in water quality and a decline in dissolved organic carbon. Carbon losses have been estimated to average around £70 per ha but could be as high as £490 per ha (2003 values from Willis et al. 2003). Changes to biodiversity are likely to include a loss of canopy resources and a possible short-term increase in saproxylic invertebrates (likely followed by a severe population decline due to a lack of suitable breeding material), although all three of the known conifer hosts are not native and endemic to Britain, so may have limited environmental value. There may, however, be an increase in ground flora (introduction of more light) particularly in hemlock stands which create dense understorey shade (Harmer et al. 2011). An average monetary value estimate for biodiversity loss is £146 per ha when estimated from Willis et al. (2003). More recent data on quantified environmental costs of the loss of mature trees are not available. Where conifer sites that are felled are scheduled for restoration under PAWS (returning Plantation sites to species mixes of Ancient Woodland Sites) with an emphasis on native broadleaf and conifer species in the mixed plantings, the result could be much greater biodiversity.
Environ –
small Small Medium Large Very
mental Impacts
Very
| ✓ |
large
30
Confidence High Confidence
Medium
Confidence
Low
Confidence
| ✓ |
Environmental impacts are assessed as medium, but main tree species affected are not natives and it is uncertain if the requirement to fell affected trees plus those in a buffer zone will continue, so a low confidence rating applies to the assessment.
Potential social impacts in the PRA area
It is likely that there would be negative social responses to damage caused by P. pluvialis plus the impacts of widespread tree felling, due to the change in visual quality of affected forests especially if they are in areas used for amenity and recreational visits, although the latter would only apply to a limited proportion of affected trees. Clearing affected, potentially dying, trees for safety would also require priority action if they were in close proximity to recreational areas.
Overall social impacts are therefore judged as likely to be medium but with a low confidence due to the lack of data on the likely extent of damage and social analysis.
Social
small Small Medium Large Very
Impacts
Very
| ✓ |
large
Confidence High Confidence
Medium
Confidence
Low
Confidence
| ✓ |
15. What is the pest’s potential as a vector of plant pathogens?
Phytophthora pluvialis is a plant pathogen with no capacity to act as a vector of other pathogens.
16. What is the area endangered by the pest?
The current findings of P. pluvialis causing disease on hemlock and Douglas fir range from the north of west Scotland (Ross-shire), Wales and down to southwest England (Cornwall). This confirms that a suitable climate for the pathogen to establish and cause disease is present throughout much of the PRA area. Phytophthora pluvialis could potentially become established throughout the ranges of its known hosts in Britain (principally Douglas fir and western hemlock) which are found in woodlands, forest, parklands and gardens (see National Forest Inventory12). Environmental conditions in the
12 National Forest Inventory https://www.data.gov.uk/dataset/ae33371a-e4da-4178-a1df 350ccfcc6cee/national-forest-inventory-woodland-england-2015
31
west of the UK are predicted to be most favourable to P. pluvialis for both establishment and spread (see Section 11).
Stage 3: Pest Risk Management
17. What are the risk management options for the UK/PRA area?
Exclusion
Recent findings indicate that P. pluvialis is already present in England, Scotland, and Wales at 35 separate locations (Section 6). It was classified by the UK Plant Health Risk Group, at least initially based on limited information about its UK distribution, as meeting the criteria to become a GB Quarantine Pest for regulatory purposes. This has enabled statutory action to be taken against findings as a precautionary measure with ongoing surveys to assess how limited or widespread P. pluvialis is and whether statutory action should be retained. Greater clarity about the extent of its distribution is also required if Protected Zone status is to be considered for parts of the UK where the pest has not been found/reported (i.e. Northern Ireland) but exclusion through a Pest Free Area from the GB area does not currently apply.
The geographical distribution of the findings also suggests that P. pluvialis has been in Britain for some time, or that there have been multiple introductions, although the level of risk identified around each pathway make this unlikely (see Section 9).
Eradication
Eradication is unlikely to be an effective option for P. pluvialis for various reasons. Eyre et al. (2022) set out some of the reasons for this which are:
• The number and widespread extent of the known outbreaks across Britain. The known extent of P. pluvialis already spans a significant area of southwest England and includes locations in Cumbria and several sites across Wales and western Scotland. In their analysis Pluess et al. (2012) found that the spatial extent of the initial infestation was significantly related to poor eradiation outcomes.
• The behaviour of the pathogen. Most Phytophthora species have highly successful survival strategies and tend to be supressed rather than eradicated by biocidal treatments (Jung et al. 2016).
• Eradication attempts with other Phytophthora introductions. There are no previous examples of non-native Phytophthora that have been found in the wider environment in the UK or possibly worldwide that have been successfully eradicated after establishment in the wider environment (Hansen 2015).
• The climatic limits of the pathogen. The presence of P. pluvialis in locations from southwest England to Northwest Scotland suggests that it is not at its climatic limits in Britain, especially as one of the outbreaks is in Surrey, England, and outside the most
32
climatically suitable areas for establishment (see Figure 3). Eradication efforts are likely to be more challenging if the pathogen is not limited by climate.
Containment and controls
As an introduced pathogen in New Zealand, P. pluvialis has not been treated as a quarantine pest but efforts have been focussed on improving understanding of pathogen behaviour and management strategies. In New Zealand, it has become clear that disease outbreaks of RNC caused by P. pluvialis are cyclical and strongly weather dependent, reoccurring at different amplitudes every 2-3 years. However, these cycles are most apparent (and possibly most damaging) in new outbreak areas and less evident in areas where the pathogen is well-established (Dick et al. 2014; Gómez-Gallego et al. 2019b). Moreover, disease cycles are not associated with tree mortality and trees recover from even severe episodes of defoliation.
If eradication in the UK is not an option, then in the short-term, management options for consideration include:
• Containment of outbreak areas. Current evidence of the potential for natural spread by P. pluvialis is limited, but the process probably operates over limited distances (a few 100s of metres; see Section 12). Logs and timber from affected trees can carry the pathogen, but available evidence suggests the risk of spread from this material is limited and likely to be slow compared to spread to infected ‘plants for planting’. Any risk of spread via harvested timber would be minimised through good biosecurity practices such as (1) jet washing machinery before it leaves affected areas to minimise the movement of contaminated/infected needles/debris through forestry operations, and (2) transfer any harvested material to already established, inspected bio-secure processors to prevent movement of infected bark/sapwood into the wood supply chain.
• Removal of affected hosts (Douglas fir and western hemlock). There is major uncertainty about the host range of P. pluvialis although preliminary testing work suggests that Scots pine, Corsican pine and Sitka spruce have little or no susceptibility. Whilst more evidence is gathered on host range (e.g. susceptibility of all UK commercially grown pine species and some broadleaf species) and on dispersal potential under UK conditions, host removal could be focussed on the two known UK hosts at the worst affected locations. Felling and clearing of the affected overstorey and associated understorey would reduce build-up of sporulating material and therefore reduce the likelihood of natural spread through aerial dispersal of P. pluvialis sporangia/zoospores. Currently, little is known about sporulation potential on the foliage of different hosts (e.g. hemlock vs Douglas fir) and how this affects natural disease spread.
• Monitoring tree recovery and assessment of episodes of disease that arise in relation to known environmental triggers. This would extend understanding of the impacts of P. pluvialis over time, for both significant outbreaks and only lightly affected outbreaks.
33
There is emerging evidence that although P. pluvialis causes bark-killing cankers on branches and main stems of both western hemlock and Douglas fir, it is not unusual for canker development to halt and recovery to occur as callus occludes areas of cambial death caused by cankers (A. Pérez-Sierra and JF Webber, unpublished data). Recovery after disease episodes may be common, and on some affected sites canker healing has been ongoing for 1-4 years. Additionally, despite confirmation of P. pluvialis at some locations, the pathogen is no longer active, only light symptoms are apparent and disease recovery is evident (A. Pérez-Sierra, unpublished data).
• Reviewing costs, impacts and benefits of host removal in relation to knowledge about host range and disease development.
Longer term options include:
• Trialling silvicultural treatments which change stand conditions, and by altering the microenvironment of infected trees reduce the likelihood of severe disease expression.
• Use of disease suppression compounds which reduce symptom expression, although such an approach would have to be consistent with certification and UKWAS. Phosphoric acid (phosphite) and copper oxychloride have both shown promise for the control of P. pluvialis in controlled inoculation experiments and within plantation systems although they have yet to be applied operationally within New Zealand forest systems. Aerial sprays of copper oxychloride are used routinely for other foliar pathogens (e.g., Dothistroma needle blight).
• Breeding for resistance as a long-term option for high value hosts. In New Zealand, field, and laboratory screening of radiata pine have shown that resistance to needle loss due to red needle cast is heritable and have identified resistance within current radiata pine breeding lines in use in New Zealand.
18. References
Abad G, Burgess T, Bienapfl JC, Redford AJ, Coffey M, Knight L. (2019). Molecular and morphological identification of Phytophthora species based on the types and other well authenticated specimens. https://idtools.org/id/phytophthora/index.php
Anon. (1965). Seed identification numbers. Forestry Commission Research Branch Paper 29, 1-370.
Boevink PC, Birch PRJ, Turnbull D, Whisson SC. (2020). Devastating intimacy: the cell biology of plant–Phytophthora interactions. New Phytologist 228, 445–458 https/doi: 10.1111/nph.16650
Brar S, Tabima JF, McDougal RL, Dupont PY, Feau N, Hamelin RC, Panda P, LeBoldus JM, Grünwald NJ, Hansen EM, Bradshaw RE & Williams NM. (2017). Genetic diversity of Phytophthora pluvialis, a pathogen of conifers, in New Zealand and the west coast of the
34
United States of America. Plant Pathology, 67, 1131–1139.
https://doi.org/10.1111/ppa.12812
Dick MA, Williams NM, Bader M-K-F, Gardner JF & Bulman LS. (2014). Pathogenicity of Phytophthora pluvialis to Pinus radiata and its relation with red needle cast disease in New Zealand. Journal of Forestry Science 44(6) https://doi.org/10.1186/s40490-014-0006-7
Dungey HS, Williams NM, Low CB & Stovold GT. (2014). First evidence of genetic-based tolerance to red needle cast caused by Phytophthora pluvialis in radiata pine. New Zealand Journal of Forestry Science, 44. https://doi.org/10.1186/s40490-014-0028-1
EPPO (2022). Global Distribution Database – Phytophthora pluvialis (PHYTUV). https://gd.eppo.int/taxon/PHYTUV/distribution Accessed 21/02/2022
Erwin DC & Ribeiro OK. (1996). Phytophthora Diseases Worldwide. American Phytopathological Society Press, St Paul, MN.
Eyre D, Everatt M, Giltrap N & Lacey K. (2022). Comments on policy options document (17 Dec 2021) for Phytophthora pluvialis from risk managers in Defra’s Risk and Horizon Scanning Team. Internal document.
Forestry Commission Plant Health (2022). Seed import data for Pseudotsuga menziesii and Tsuga heterophylla. Unpublished report. Or
Fraser S, Gomez-Gallego M, Gardner J, Bulman LS, Denman S & Williams NM. (2020). Impact of weather variables and season on sporulation of Phytophthora pluvialis and Phytophthora kernoviae. Forest Pathology, 50(n/a), e12588. https://doi.10.1111/efp.12588
Ganley RJ, Williams NM, Rolando CA, Hood IA, Dungey HS, Beets PN, Bulman LS. (2014). Management of red needle cast, caused by Phytophthora pluvialis, a new disease of radiata pine in New Zealand. New Zealand Plant Protection 67:48–53. Available from: http://www.nzpps.org/nzpp_abstract.php?paper=670480
Gardner J, Dobbie K, Fraser S. (2020). Shake it: how to collect Douglas fir needles for Phytophthora diagnostics. Forest Health News – Scion, No. 300, May 2020, ISSN 1175- 9755
Govers LL, Man in ‘t Veld WA, Meffert JP, Bouma TJ, van Rijswick PCJ, Heusinkveld JHT, Orth RJ, van Katwijk MM, van der Heide T. (2016). Marine Phytophthora species can hamper conservation and restoration of vegetated coastal ecosystems. Proc. R. Soc. B 283: 20160812. http://dx.doi.org/10.1098/rspb.2016.0812
Gómez-Gallego M, Bader MK, Scott PM, Leuzinger S, Williams NM. (2017). Phytophthora pluvialis studies on Douglas-fir require Swiss needle cast suppression. Plant Disease 101(7),1259-1262. https://doi: 10.1094/PDIS-12-16-1738-RE
Gómez-Gallego M, Gommers R, Bader MK-F, Williams NM. (2019a). Modelling the key drivers of an aerial Phytophthora foliar disease epidemic, from the needles to the whole plant. PLoS ONE 14(5): e0216161
Gómez-Gallego M, LeBoldus JM, Bader M K-F, Hansen E, Donaldson L & Williams NM. (2019b). Contrasting the pathogen loads in co-existing populations of Phytophthora
35
pluvialis and Nothophaeocryptopus gaeumannii in Douglas fir Plantations in New Zealand and the Pacific Northwest United States. Phytopathology 109(11)
https://doi.org/10.1094/PHYTO-12-18-0479-R
Hansen E. (2015). Phytophthora Species Emerging as Pathogens of Forest Trees. Current Forestry Reports 1. https://doi:10.1007/s40725-015-0007-7
Hansen EM, Reeser P, Sutton W, Gardner J, Williams N. (2015). First report of Phytophthora pluvialis causing needle loss and shoot dieback on Douglas-fir in Oregon and New Zealand. Plant Disease 99, 727.
Hansen E, Bourret T, Kanaskie A, LeBoldus L, Navarro S, Reeser P & Sutton W. (2017a). Current status of Phytophthora pluvialis in western North America. Proceedings of the 8th Meeting of the International Union of Forestry Research Organisations IUFRO Working Party S07-02-09 Phytophthora in Forests and Natural Ecosystems, Hanoi-Sapa Viet Nam March 2017 http://forestphytophthoras.org/sites/default/files/proceedings/IUFRO-WP-7-02- 09-Sapa-2017-abstracts-V2.pdf page 24
Hansen EM, Reeser PW & Sutton W. (2017), Ecology and pathology of Phytophthora ITS clade 3 species in forests in western Oregon, USA, Mycologia 109(1), 100-114 http://doi: 10.1080/00275514.2016.1273622
Harmer R, Beauchamp K & Morgan G. (2011). Natural regeneration in western hemlock plantations on ancient woodland sites. Research Note 11: Forestry Commission, Edinburgh. https://www.forestresearch.gov.uk/documents/295/FCRN011_ShIKdPO.pdf
Hood I, Fraser S, Husheer S, Gardner J, Evanson T, Tieman G, Banham C & Wright L. (2022). Infection period of Phytophthora pluvialis and Phytophthora kernoviae in relation to weather variables in Pinus radiata forests in New Zealand. Submitted to journal.
Jung T et al. (2016). Widespread Phytophthora infestations in European nurseries put forest, semi-natural and horticultural ecosystems at high risk of Phytophthora diseases. Forest Pathology 46 (2):134-163. https://doi.org/10.1111/efp.12239
Pérez-Sierra A, Chitty R, Eacock A, Jones B, Biddle M, Crampton M, Lewis A, Olivieri L & Webber J. (2022). First report of Phytophthora pluvialis in Europe causing resinous cankers on western hemlock. New Disease Report, https://doi.org/10.1002/ndr2.12064
Pérez-Sierra A, Jones B, Biddle M & Webber J. (2022b). Phytophthora pluvialis – a new threat to forestry? Quarterly Journal of Forestry 116(2), 128-130.
Pluess T, Cannon R, Jarosik V, Pergl J, Pysek P & Bacher S. (2012). When are eradication campaigns successful? A test of common assumptions. Biological Invasions 14 (7):1365-1378. doi: 10.1007/s10530-011-0160-2.
Prochazkova Z & Jancarek V. (1991). Diseases in Czech-Slovak forest nurseries. In Proceedings of the first meeting of IUFRO working party S2.07-09 (Diseases and insects in forest nurseries, Pp. 37–50). (J.R. Sutherland and S. Glover, eds.). Inform. Rep. BC-X 331, Pacific For. Centre, For. Canada, Victoria, BC.
Reeser P, Sutton W & Hansen E. (2013). Phytophthora pluvialis, a new species from mixed tanoak-Douglas-fir forests of western Oregon, U.S.A. North American Fungi 8, 1–8. https://doi.org/10.2509/naf2013.008.007
36
Reeser P, Sutton W, Ganley B, Williams N, Hansen E. 2015. Phytophthora pluvialis. Forest Phytophthoras 5(1) https://doi.10.5399/osu/fp.5.1.3745
Scion (2022). Phytophthora pluvialis outbreak parameters in New Zealand. Unpublished report.
Scott PM, Taylor P & Williams N. (2019). Contrasting the infection and survival of Phytophthora pluvialis and Phytophthora cinnamomi in Pinus radiata roots. Australasian Plant Pathology https://doi.10.1007/s13313-019-0619-7
Strong RA, Kolodny E, Kelsey RG, Gonz~lcz-Hernfindez MP, Vivanco JM &¯Manter DK. (2013). Effect of plant sterols and tannins on Phytophthora ramorum growth and sporulation. Journal of Chemical Ecology 39,733 743 https://doi.10.1007/s10886-013- 0295-y
Webber JF. (2010). Pest risk analysis and invasion pathways for plant pathogens. New Zealand Journal of Forestry Science 40, 45-56.
Webber J. (2022). Phytophthora ramorum – a developing story. Quarterly Journal of Forestry 116(2), 123-127.
Wang W, Liu X & Govers F. (2021). The mysterious route of sterols in oomycetes. PLOS Pathogens
https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009591
Williams N & Hansen E. (2018). Red needle cast. In: Compendium of Conifer Diseases, Second Edition. (eds Hansen EM, Lewis KJ and Chastagner GA), pages 16-18. APS publications.
Willis K, Garrod G, Scarpa R, Powe N, Lovett A, Bateman I, Hanley N & Macmillan D. (2003). The social and environmental benefits of forests in Great Britain. Forestry Commission Report. 36pp. 2003. Edinburgh.
Name of Pest Risk Analysts(s)
Joan Webber, Forest Research
Joan.webber@forestresearch.gov.uk

© Crown copyright 2022
37
You may re-use this information (excluding logos) free of charge in any format or medium, under the terms of the Open Government Licence v.2. To view this licence visit www.nationalarchives.gov.uk/doc/open-government-licence/version/2/ or email PSI@nationalarchives.gov.uk
This publication is available via the UK Plant Health Information portal https://planthealthportal.defra.gov.uk/
This PRA has been undertaken following IPPC International Standards for Phytosanitary Measures (ISPMs 2 and 11) and it provides technical evidence relating to the risk assessment and risk management of this pest.
Any enquiries regarding this publication should be sent to us at
The Chief Plant Health Officer
Department for Environment, Food and Rural Affairs
Room 11G32
Sand Hutton
York
YO41 1LZ
Email: plantpestsrisks@defra.gov.uk
38